Ice in the polar regions even has an influence high up in the sky: ice crystals in clouds are part of the process which creates the Arctic and Antarctic ozone holes.
Ozone is 3 atoms of oxygen. High up in the stratosphere, above the region where most planes fly, ozone protects earth from harmful ultraviolet radiation. Stratospheric ozone serves as a welcome planetary "sun-block."
|
|
|
But in the 1980's, research here in Antarctica showed that chlorine from chemicals called "chlorofluorocarbons", CFCs for short, were destroying the ozone, and creating what became known as a "hole."
Further research showed that CFCs-then found in aerosol sprays and refrigerators-were interacting in unique ways with the ice crystals that made up "polar stratospheric clouds"-high, cold, and a perfect place for this kind of chemistry. Incoming radiation freed up the chlorine, which then interacted with ozone, breaking it down. In further reactions, the chlorine-like some chemical Pac Man-could destroy up to 100,000 ozone molecules!
|
As we saw in PASSPORT TO ANTARCTICA, program 8, research on green plants on the Antarctic Peninsula, and on the creatures living in the southern ocean, are showing biological consequences of increased levels of ocean.
In 1987, Earth's nations came together to ban CFCs by a treaty known as the "Montreal Protocol," and CFC emissions have been going down.
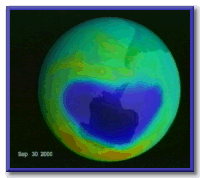
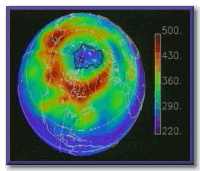
Still the year 2000 ozone hole over Antarctica was a record. NASA's TOMS satellite showed that the hole reached nearly 30 million square kilometers, 3 times larger than the entire land mass of the United States.
Image courtesy of NASA
Ozone levels over the Arctic have also declined 50%.
Nevertheless most researchers think future holes should decline in size and severity.
The kind of international cooperation which characterizes research in Antarctica may well prove to be the way to help the world.
Websites
• Antarctic Ozone Hole Research
Background information on the ozone hole, what chemical causes it, and why the discovery of the thinning ozone layer was important.
• Ozone
Images and animations of the ozone hole from 1998 to 2000.
• 2001 Ozone Hole About the Same Size as Past Three Years
NASA and NOOA scientists have discovered that this year's ozone hole is similar in size to the past three years.
• NOAA, NASA Report 2001 Ozone Hole Similar in Size to Holes of Past Three Years
Article on the Antarctic ozone hole explaining that scientists see a leveling off in the size of the ozone hole
• Climate Monitoring & Diagnostics Laboratory South Pole Ozone Program
The most recent ozone hole information including tracking the current ozone hole
• Science: The Antarctic Ozone Hole
Background information on the Antarctic ozone hole
• The Science of Stratospheric Ozone
The basics of ozone, ozone depletion and information on the Antarctic ozone hole
• Total Ozone Mapping Spectrometer (TOMS)
TOMS home page where you will find information and data from the TOMS instruments.
• Today's Ozone
Latest images on the ozone hole.
![]()

